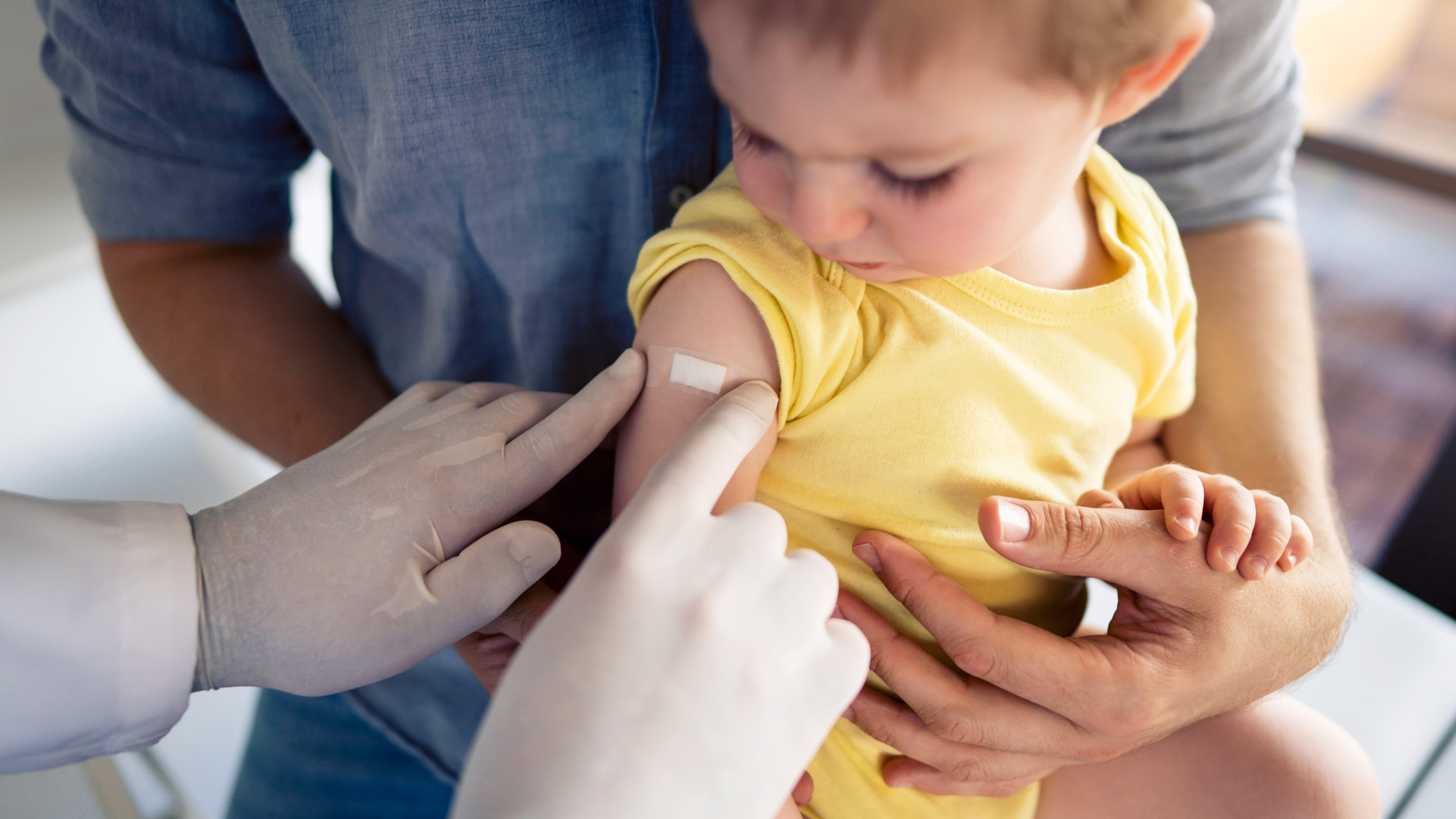
Immuno-persistence of the different primary polio vaccine schedules and immunogenicity of the booster dose by sabin inactivated or bivalent oral poliovirus vaccine in children aged 4 years: an open-label, randomised, controlled phase

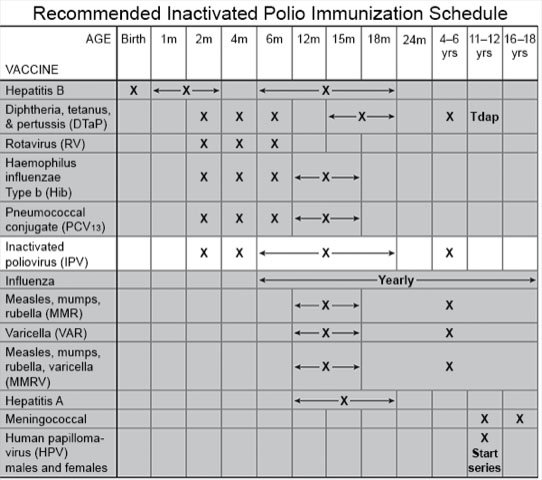
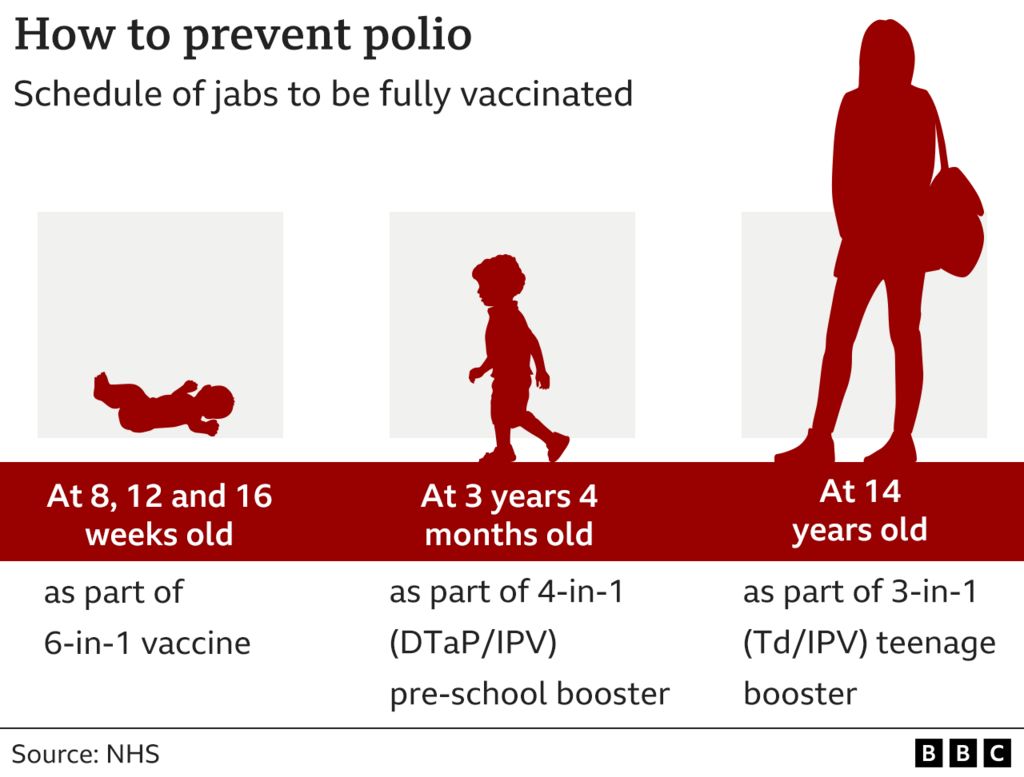
VUCA Health - In the United States, inactivated polio vaccine (IPV) is recommended. We are no longer using oral polio vaccine (OPV). Reasons for not using OPV are related to the paralytic


:max_bytes(150000):strip_icc()/poliovax-fd552b4321d94c89af4836f3e73431fa.jpg)